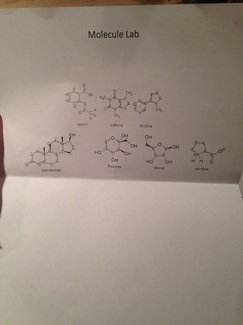
During this lab, we drew Lewis dot structures for multiple atomic compounds, organic and inorganic. After we had practice, we made 3D models for some of them out of gumdrops, raisins, and toothpicks.
From this lab, I know how to look at the periodic table of elements and identify how many valence electrons a given element has based off of the column number it is in from left to right. From this I learned how to connect each element in a compound using Lewis dot structures.
I now know the fine details of the shape of the molecules and how to display them using Lewis dot structures. As well of this I learned how about the bonds that connect them, and how they relate to one another.
From this lab I can conclude that patterns in molecular shapes can help identify deeper similarities among compounds.
From this lab, I know how to look at the periodic table of elements and identify how many valence electrons a given element has based off of the column number it is in from left to right. From this I learned how to connect each element in a compound using Lewis dot structures.
I now know the fine details of the shape of the molecules and how to display them using Lewis dot structures. As well of this I learned how about the bonds that connect them, and how they relate to one another.
From this lab I can conclude that patterns in molecular shapes can help identify deeper similarities among compounds.










 RSS Feed
RSS Feed
