Before leading us through an experiment that would observe how magnesium reacts with citric acid, Andrew introduced us to atoms and what they're all about. To start, atoms are the smallest unit of any element, they have properties of the element which they create. Atoms are made of three subatomic particles. Protons, electrons, and neutrons. Though protons are positive and neutrons are neutral, they both have a significant mass. This means that they add something to the atom that is prominently detectable. Electrons though, have an insignificant mass. Andrew used an example that stuck with me to explain this, having an insignificant mass is like bringing a feather on to a scale and seeing if it noticebly changes your weight. It doesn't.
Atoms have different energy levels that surround the nucleus, this is the center of an atom, and a cell in general. Protons and neutrons are located inside of the nucleus. Electrons though, orbit the nucleus in the varying energy levels.
The info graphic I've made below shows how to identify atom looks like for an element, on the periodic table.
Atoms have different energy levels that surround the nucleus, this is the center of an atom, and a cell in general. Protons and neutrons are located inside of the nucleus. Electrons though, orbit the nucleus in the varying energy levels.
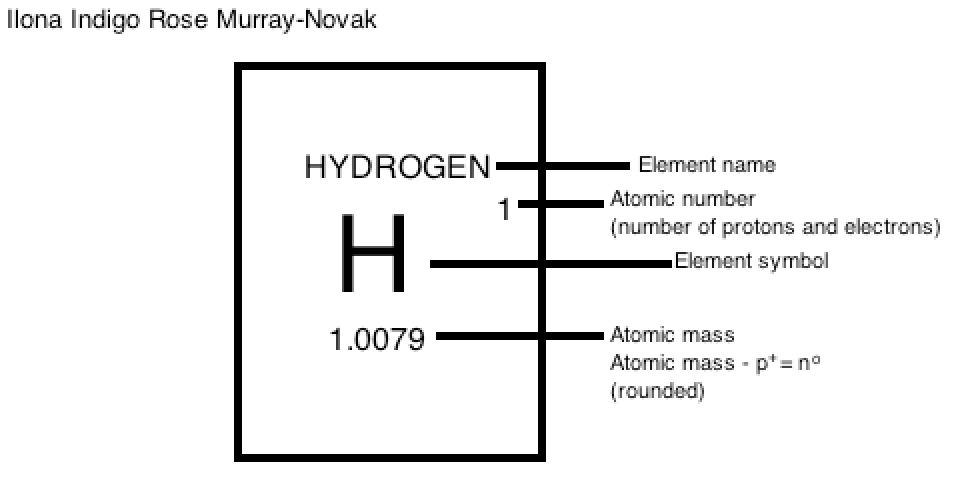
The info graphic I've made below shows how to identify atom looks like for an element, on the periodic table.
Todays experiment is about watching what happens when magnesium and citric acid atoms combine. When they combine, whats happening is that one atom is giving another an electron(s). They do this because every atom wants a full set of electrons. Taking a big beaker and filling it with distilled water and magnesium strips, my group then added IN citric acid through a funnel and quickly put a balloon on top. The balloon would seal in the hydrogen being created by this reaction. From observing the solution we could see that the magnesium was dissolving and that it was turning into a gas, expanding the balloon. The glass was warm to the touch because of the energy that the reaction was creating. When the fizzing had subsided, Andrew tied off the balloon, it floated! While we were playing with it, we accidentally let it go and Andrew had to use a big stick with tape on the end to get it off of the ceiling. To end the experiment, we turned the lights off and held each balloon on the end of a long stick. They each exploded very loudly with a burst of fire. Very cool and unexpected results from some metal and some sour stuff!



 RSS Feed
RSS Feed
